What is the MOTS-c peptide?
Last reviewed: · Reviewed by the mots-c.com Editorial Team
MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a 16-amino-acid peptide encoded directly within mitochondrial DNA. It acts as a signaling molecule that helps the body regulate metabolism, respond to stress, and maintain energy balance — particularly during exercise and caloric restriction.
A peptide made by mitochondria
For most of modern biology, mitochondria were considered passive cellular power plants. The discovery of MOTS-c in 2015 changed that. Researchers found that mitochondria themselves encode small peptides — called mitochondrial-derived peptides (MDPs) — that act as messengers between cells and organs.
MOTS-c is the most studied of these. It is produced inside mitochondria, released into circulation, and exerts effects on skeletal muscle, fat tissue, the liver, and the brain.
How MOTS-c works in the body
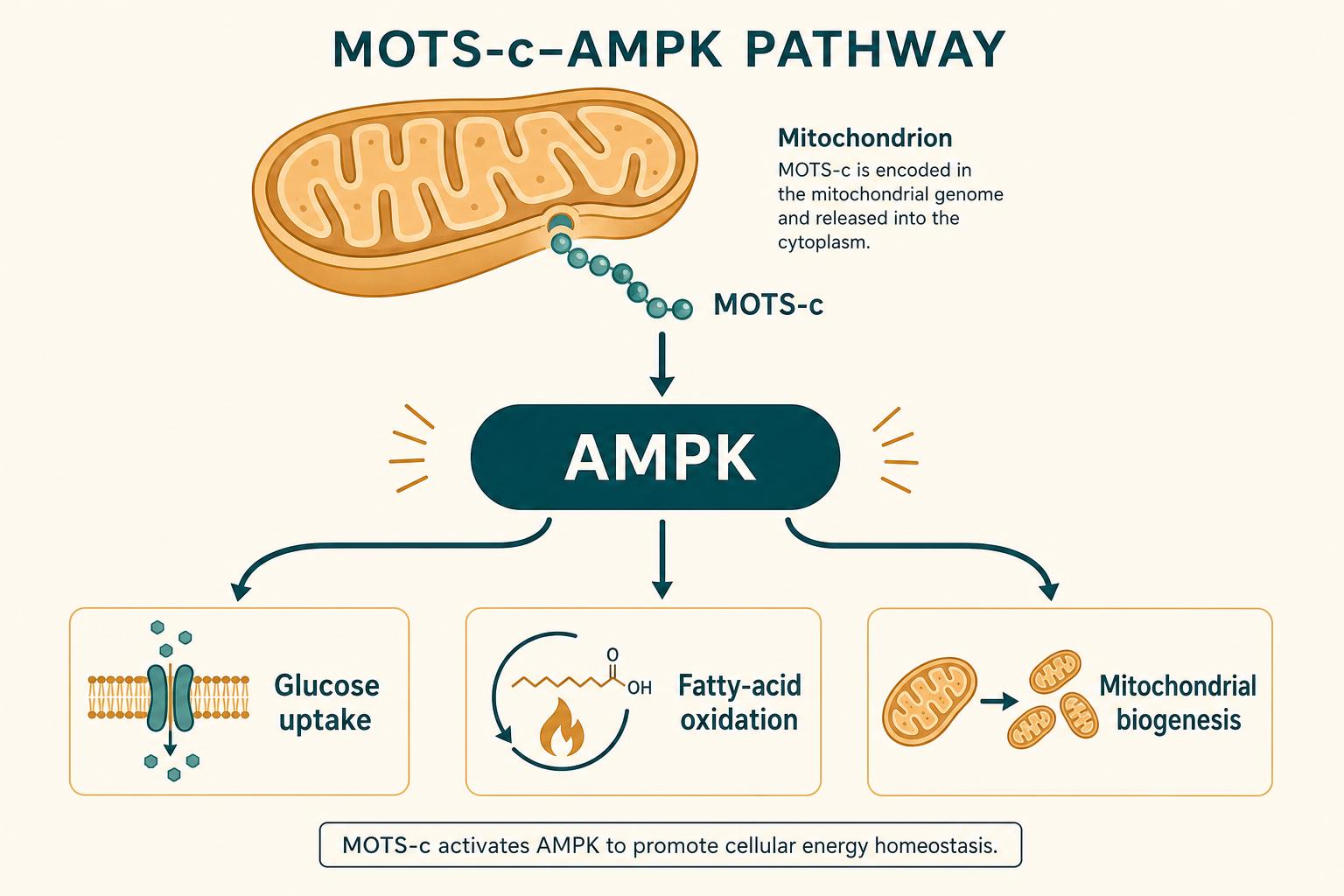
MOTS-c primarily activates AMPK (AMP-activated protein kinase), the master sensor of cellular energy. When AMPK is activated, the body:
- Increases glucose uptake into muscle cells
- Burns fatty acids for energy more efficiently
- Improves insulin sensitivity
- Triggers mitochondrial biogenesis (more, healthier mitochondria)
This is the same pathway activated by exercise and by metformin — making MOTS-c often described as an "exercise-mimetic" peptide in the research literature.
How MOTS-c compares to other longevity signals
MOTS-c is often grouped with other "exercise-mimetic" or metabolic-health interventions. It is not interchangeable with them, but the comparison helps locate it on the map:
- Exercise. Endurance training raises endogenous MOTS-c. Some of the metabolic benefits of exercise may be downstream of mitochondrial-derived peptide release.
- Metformin. Like MOTS-c, metformin activates AMPK, but indirectly via the respiratory chain. MOTS-c appears to act more directly on AMPK regulators.
- GLP-1 agonists. These work on appetite and insulin secretion; MOTS-c works on cellular energy handling. Different lever, sometimes overlapping outcomes.
- Other MDPs. Humanin and SHLPs share the mitochondrial-encoded origin but signal differently. MOTS-c is the most studied of the family.
Why levels matter
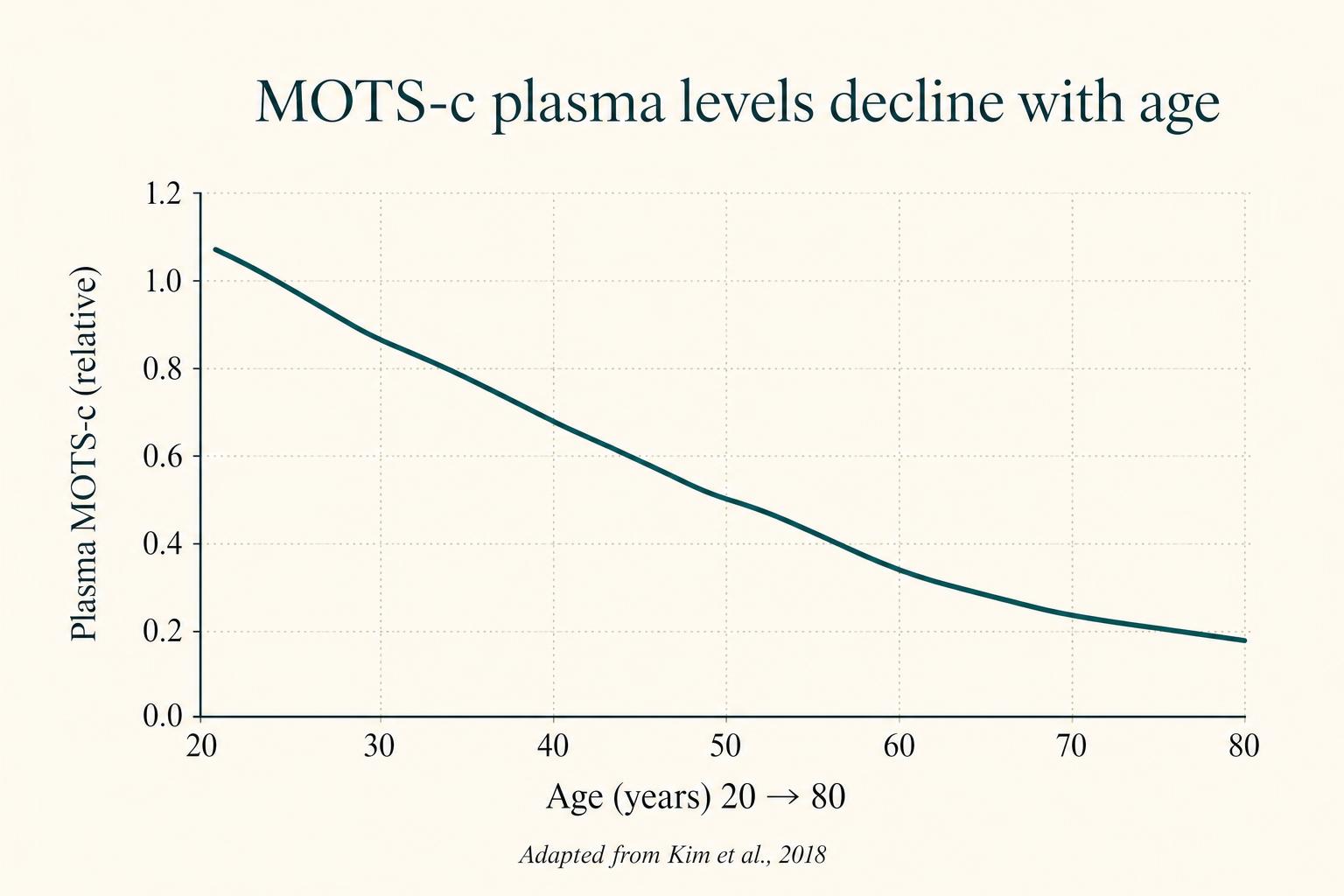
MOTS-c levels in human plasma decline with age. They are also lower in people with type 2 diabetes and obesity. This correlation has driven interest in MOTS-c as a potential intervention for metabolic disease and healthy aging.
Where MOTS-c is studied
The first wave of MOTS-c research focused on metabolism and obesity. Subsequent work has extended into:
- Skeletal muscle. Exercise capacity, sarcopenia prevention, mitochondrial density.
- Adipose tissue. Fat oxidation, browning of white fat, protection against diet-induced obesity.
- Liver. Hepatic insulin sensitivity, NAFLD-relevant lipid handling.
- Bone. Osteoblast support, with implications for age-related bone loss.
- Cardiovascular tissue. Endothelial function and ischemia-reperfusion protection in early models.
Molecular details
MOTS-c is encoded within the mitochondrial 12S rRNA region using a non-standard mitochondrial genetic code. The mature peptide is 16 amino acids long, with the human sequence reported as MRWQEMGYIFYPRKLR. Once translated, it can act inside the cell of origin or be secreted into circulation to act on distant tissues.
Under metabolic stress — for example, glucose deprivation or exercise — MOTS-c translocates from mitochondria into the nucleus, where it modulates the expression of stress-response and antioxidant genes (notably AMPK-linked transcriptional programs). This dual role, both as a circulating signaling peptide and as a nuclear regulator, is unusual and helps explain its broad effects.
Cellular mechanisms in detail
- Glucose handling. Increases GLUT4 translocation to muscle cell membranes, raising insulin-independent glucose uptake.
- Folate / one-carbon metabolism. Inhibits the folate cycle, increasing AICAR, which directly activates AMPK.
- Lipid oxidation. Shifts substrate preference in skeletal muscle from glucose toward fatty acids during sustained work.
- Mitochondrial quality. Promotes biogenesis of new, more efficient mitochondria via PGC-1α-related signaling.
- Stress response. Engages nuclear antioxidant programs (NRF2/ARE-related) under metabolic stress.
Clinical relevance
The clinical significance of MOTS-c rests on three observations consistently reported in peer-reviewed work:
- Plasma MOTS-c is lower in people with type 2 diabetes, obesity, and advancing age.
- Endogenous MOTS-c rises with exercise, suggesting it mediates part of exercise's metabolic benefit.
- Exogenous MOTS-c improves insulin sensitivity, body composition, and exercise capacity in animal models.
These observations are what motivate ongoing translational research, but they do not establish MOTS-c as a proven human therapy. See the key studies page for the full evidence base.
What's next
MOTS-c is still firmly in the research phase. The next pages cover what current studies actually show about benefits, what's known about safety and dosing, and the key studies behind it.